اثرات اسید فسفریک بر اکوسیستم های آبی
اثرات اسید فسفریک بر اکوسیستم های آبی
اثرات اسید فسفریک بر اکوسیستم های آبی ، را می توان نوعی اثر منفی در نظر گرفت. اسید فسفریک یک اسید قوی نیست. از اسید استیک قوی تر است، اما از اسید سولفوریک و اسید کلریدریک ضعیف تر است. نمک های فسفریک اسید را می توان با جایگزینی یک، دو یا سه یون هیدروژن تشکیل داد. به عنوان مثال، NaH2PO4 ، سدیم دی هیدروژن فسفات، می تواند با واکنش یک مول اسید فسفریک با یک مول هیدروکسید سدیم تشکیل شود.
اثرات بر اسید فسفریک بر اکوسیستم های آبی
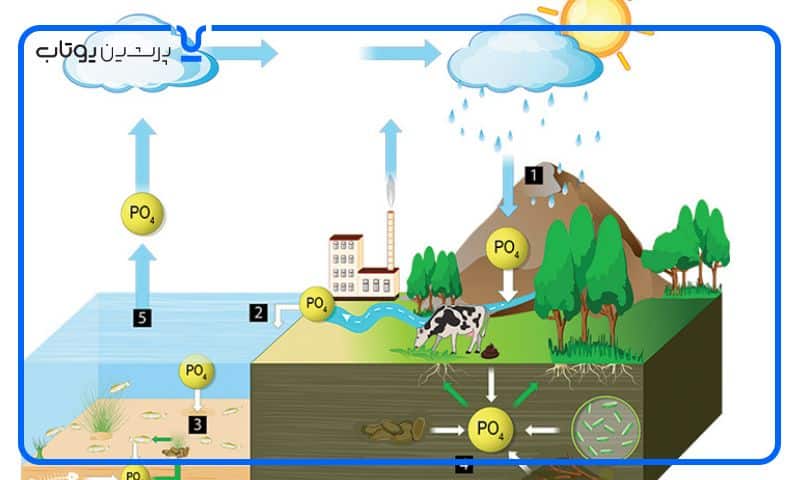
افزایش غلظت فسفات در آب های سطحی نیز رشد حیات گیاهان آبزی را افزایش می دهد. رواناب از زمین های کشاورزی بارور می تواند رشد گیاهان را در دریاچه ها و رودخانه ها تحریک کند. در چنین مواردی، غلظت اسید فسفریک تجمع یافته در فاضلاب باعث ایجاد اتروفیزیک، به معنی رشد سریع جلبک ها می شود که می تواند به آبزیان آسیب برساند. بسیاری از مقامات محلی برای کاهش خطر eutrophication (اتروفیکاسیون، فرایندی است که طی آن یک کل آب یا قسمت هایی از آن به تدریج با مواد معدنی و مواد مغذی غنی می شود.)دریاچه، استفاده از فسفات ها را در مواد شوینده ممنوع کرده اند. با این حال، هنگامی که در یک محیط کنترل شده استفاده می شود، اسید فسفریک عملکرد خوبی را ایجاد می کند و آسیبی به اکوسیستم وارد نمی کند.
آیا اسید فسفریک برای محیط زیست مضر است؟
اسید فسفریک جزء اکثر کودها است که به رشد گیاهان کمک می کند. هنگامی که بیش از حد اعمال می شود یا در زمان اشتباه اعمال می شود، مانند قبل از باران، بیشتر آن شسته می شود و به آبراهه های محلی ختم می شود. این نوع آلودگی را آلودگی منبع غیر نقطه ای می نامند. این باعث اتروفیکاسیون (کاهش اکسیژن محلول در آبها ناشی از افزایش مواد معدنی و مواد مغذی آلی) رودخانه ها و دریاچه ها می شود. این کاهش سطح اکسیژن در آب باعث خفه شدن ماهی می شود. شهرداری بعضی از شهرها استفاده از کودهای حاوی اسید فسفریک را برای چمن ممنوع کرده اند. این قوانین برای حفاظت از کیفیت آب محلی در دریاچه ها، نهرها و حوضچه ها طراحی شده است. به گفته چندین متخصص، در بیشتر موارد فسفر برای حفظ چمن سالم لازم نیست.
متابولیسم فسفر موجودات صخره ای با همزیستی جلبک
فسفر (P) ، یک جزء اساسی ساختاری و عملکردی از همه موجودات زنده، به عنوان آخرین ماده مغذی محدود کننده در اکوسیستم های دریایی در نظر گرفته می شود. برای بهینه سازی جذب، گونه های دریایی مانند تک یاخته ها، اسفنج ها، روزن داران، صدف ها و مرجان های صخره ای، با جلبک ها رابطه ای همزیستی برقرار کرده اند، که مواد زائد میزبان حیوانات را بازیافت کرده و مواد مغذی معدنی محلول را به مولکول های آلی تبدیل می کند. در دسترس میزبان آنها چنین ارتباطاتی در محیطی که دسترسی به مواد مغذی در آن کم است، مزیت رقابتی ایجاد می کند.
استفاده از ضایعات در صنعت اسید فسفریک از طریق توسعه فرآیندهای سازگار با محیط زیست برای طیف وسیعی از محصولات حاوی فسفر
یک پروژه با بودجه اتحادیه اروپا و یک استراتژی ابتکاری برای به حداقل رساندن ضایعات در صنعت اسید فسفریک ایجاد شده است.
پروژه “استفاده از زباله در صنعت اسید فسفریک از طریق توسعه فرایندهای سازگار با محیط زیست و سازگار با محیط زیست” (Ecophos) بر تعدادی از تکنیک های جدید برای تولید نمک های فسفر، اسید فسفر و فسفات متمرکز شده است. هدف توسعه فناوری فرآیند صنعتی منابع و انرژی برای تولید طیف وسیعی از مواد حاوی فسفر بود. کنسرسیوم روش های فعلی را برای استفاده و به حداقل رساندن ضایعات جامد صنعتی ناشی از تولید اسید فسفریک بهبود بخشید. تولید نسل جدید کودهای فسفری نیز مورد مطالعه قرار گرفت. از مدل های ریاضی و ابزارهای مهندسی مبتنی بر کامپیوتر برای تضمین عملکرد کارآمد و سازگار با محیط زیست سیستم تولید استفاده شد. هدف کاهش هزینه، اتلاف و مصرف انرژی بود. سیستم تولید با توجه به خواص زباله و پتانسیل کاهش اثرات زیست محیطی و افزایش پایداری طبقه بندی می شود. یک سیستم متخصص کاربران را قادر می سازد تا طرح تولیدی را که نزدیک به نیازهای خود است انتخاب کنند. ابتکار Ecophos یک فناوری فرآیند صنعتی دوستدار محیط زیست تولید کرد که می تواند هم منابع و هم انرژی را حفظ کند.





1 Comment
حفظ محیط زیست توسط ما انسان ها در این کم آبی بسیار حائز اهمیت هست