اکسیداسیون اسید اسکوربیک
اکسیداسیون اسید اسکوربیک
عوامل موثر در اکسیداسیون اسید اسکوربیک در این مقاله مورد بررسی قرار گرفته است.
در مواد غذایی حاوی اسید اسکوربیک، تا زمانی که اسید اسکوربیک و اسید دهیدرواسکوربیک (مولکول تشکیل شده در اثر اکسیداسیون آسکوربات، اسید دهیدرواسکوربیک است.)، به یکدیگر واکنش نشان دهند، هیچ ویتامین C از بین نمی رود. با این حال، اسید دهیدرواسکوربیک می تواند در واکنش های برگشت ناپذیر واکنش بیشتری نشان دهد. اگر این اتفاق بیفتد، واقعاً ویتامین C از بین می رود.
به طور کلی، تولید کنندگان مواد غذایی و مصرف کنندگان نمی خواهند ویتامین C قبل از اینکه بتوانیم آن را مصرف کرده و عملکرد آن در بدن ما انجام شود، از بین برود. بنابراین، دانشمندان مواد غذایی، نحوه کنترل میزان اکسیداسیون اسید اسکوربیک و همچنین تجزیه اسید دهیدرواسکوربیک را مورد بررسی قرار داده اند. پنج پارامتر تأثیرگذار پیدا شده است.
پارامترهای تاثیر گذار بر اکسیداسیون اسید اسکوربیک
اکسیژن، یون های فلزی، PHآب میوه، نور و دما
اکسیداسیون اکسیژن و اسید اسکوربیک
در صورت عدم وجود اکسید کننده، می توان اکسیداسیون اسید اسکوربیک را کند یا از آن جلوگیری کرد. به عبارت دیگر، باید یک مولکول وجود داشته باشد که الکترون ها را دریافت کند. اکسیژن به عنوان یک اکسید کننده بسیار خوب شناخته شده است. در واقع، نام “اکسید کننده” از اکسیژن گرفته شده است، زیرا اکسیژن یکی از اولین اکسید کننده های کشف شده است.
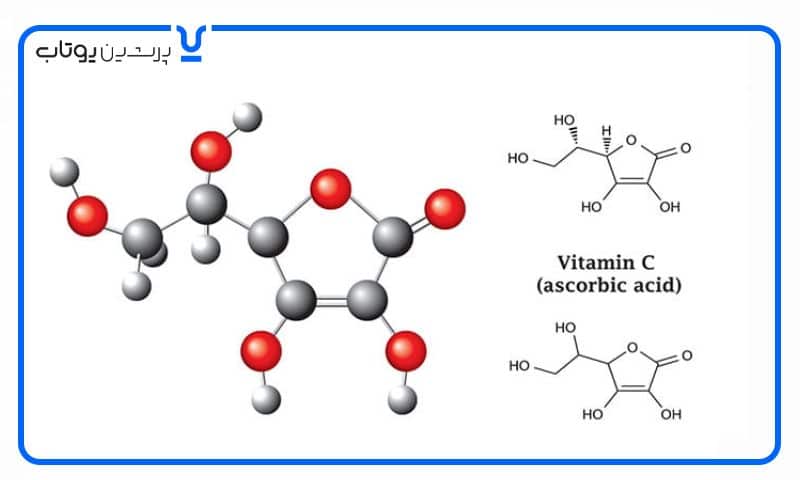
وقتی اسید اسکوربیک در هوای آزاد که حاوی اکسیژن است باز بماند، مستعد اکسیداسیون است. بنابراین، در صورت عدم وجود اکسیژن، اکسیداسیون اسید اسکوربیک کمتر رخ می دهد. ممکن است اکسید کننده های دیگری نیز وجود داشته باشند، اما حذف این ماده مهم تأثیر بسزایی خواهد داشت.
تولیدکنندگان این مشکل را با هواگیری آب میوه حل می کنند. با هوادهی آب، بیشتر اکسیژن از آب خارج می شود. پس از هوادهی تا آنجا که ممکن است هوای کمی در بطری باقی می ماند. بنابراین، آنها اغلب در سطح بالایی پر می شوند.
آب پرتقال را در خانه در یک بطری در بسته نگهداری کنید و بعد از استفاده حتما در آن را ببندید. این قضیه، میزان اکسیژن ورودی به آب پرتقال و اکسیداسیون اسید اسکوربیک را محدود می کند.
یون های فلزی و واکنش های احیا
یون های فلزی در مشارکت در اکسیداسیون و عکس العمل های احیایی آن نیز خوب عمل می کنند. در مورد اسید اسکوربیک، یون های فلزی می توانند واکنش را کاتالیز کنند. این به این معنی است که آنها در طول واکنش عادت نمی کنند، اما می توانند به عنوان محل ذخیره موقت الکترون ها عمل کنند.
یک توصیه عملی در این زمینه وجود دارد، بهتر است آب پرتقال را در ظرف مسی ذخیره نکنید. یون های مس واکنش را تا حد زیادی تسریع می کنند.
PHبر اکسیداسیون بسیار تأثیر می گذارد.
برای بسیاری از واکنش ها، pH بر سرعت واکنش تأثیر می گذارد. برای واکنش هایی که شامل یک جزء اسیدی یا قلیایی هستند، این امر به ویژه مهم است.
در واکنش اکسیداسیون اسید اسکوربیک، دیدید که پروتون (H + ) و الکترون (e – ) آزاد می شود. اما اگر اسید اسکوربیک در محیطی بسیار اسیدی قرار گیرد، پروتون های زیادی در اطراف آن وجود دارد. این می تواند واکنش را کند، کند. مشخص شده است که واکنش سریعتر در pH 4 انجام می شود.

در pH بالاتر (قلیایی تر) یا پایین تر (اسیدی تر)، واکنش کندتر پیش می رود. از آنجا که pH یک پرتقال در حدود 3-4 است، آب پرتقال در واقع مکان مناسبی برای بروز این واکنش است.
تا زمانی که پرتقال کامل باشد، هیچ واکنشی نشان نمی دهد، در برابر اکسیژن و نور محافظت می شود. اما هنگامی که شروع به تکه کردن و آب گرفتن پرتقال می کنید، اکسیداسیون اتفاق می افتد.
اکسیداسیون نور و ویتامین C
مشخص است که نور بر میزان اکسیداسیون ویتامین C تأثیر می گذارد، مانند اکسیداسیون روغن زیتون.
هر چند تأثیر دقیق نور پیچیده است و مکانیسم دقیق آن هنوز مشخص نیست. اما توصیه کلی این است که آب پرتقال را در مکانی تاریک نگه دارید.
دما و سرعت واکنش
دما حرکت مولکول ها را در غذا تعیین می کند. هرچه درجه حرارت بالاتر باشد، اتم ها و مولکول های بیشتری در اطراف حرکت می کنند. این امر باعث می شود مولکول ها در نزدیک یکدیگر باشند و در نتیجه واکنش نشان دهند. بنابراین، دمای بالاتر اغلب منجر به سرعت واکنش بیشتر می شود. این مورد در مورد اکسیداسیون ویتامین C نیز صادق است، همانطور که توسط محققان در آب توت فرنگی بررسی شده است .
واکنش میلارد ویتامین C
هنگامی که ویتامین C اکسیده شد، می تواند واکنش های بیشتری را در مسیرهای مختلف نشان دهد. یکی از آنها واکنش میلارد است. واکنش میلارد از طریق مجموعه ای از واکنش های پیچیده باعث قهوه ای شدن بسیاری از محصولات غذایی می شود .
علاوه بر واکنش میلارد، راه های مختلفی برای اسید دهیدرواسکوربیک وجود دارد که می تواند واکنش بیشتری نشان دهد، که بحث بیشتر در مورد آنها در این مقاله نمی گنجد. همچنین، تلاش برای جلوگیری از اکسیداسیون اسید اسکوربیک احتمالاً راهی بهتر از، پیشگیری از واکنش های دیگر است. اسید دهیدرواسکوربیک به سادگی پایدارتر است و به احتمال زیاد کنترل آن سخت تر است.
پس:
اگر نمی خواهید دچار اسکوربوت شوید، لطفاً آب پرتقال و محصولات حاوی اسید اسکوربیک را، در یک ظرف مسی، در یک مکان گرم، با اکسیژن زیاد و در نور زیاد خورشید نگهداری نکنید.




