Citric acid cycle reactions
Citric acid cycle
It is a Citric acid cycle reactions set of chemical reactions used by all aerobic organisms to produce energy through the oxidation of acetate from carbohydrates, fats, and proteins to carbon dioxide.
Citric acid cycle reactions
Reaction 1: Synthesis of citric acid
Acetyl CoA and oxalactic acid condense to form citric acid. The acetyl group CH3COO is transferred from CoA to oxaloacetic acid in ketone carbon and then to alcohol. The net effect is to attach a 2-carbon component to a 4-carbon component and produce citric acid, which is 6 carbons. This is just the synthesis of citric acid. This reaction is catalyzed by citric acid synthetase.
Reaction 2: Dehydration of an alcohol
Two steps are required to isomerize the -OH group position on citric acid. This is the first stage of dehydration of alcohol to make an alkene. Cis aconitic acid remaining in preparation for the next step required to bind the enzyme aconitase. This reaction is catalyzed by aconitase.
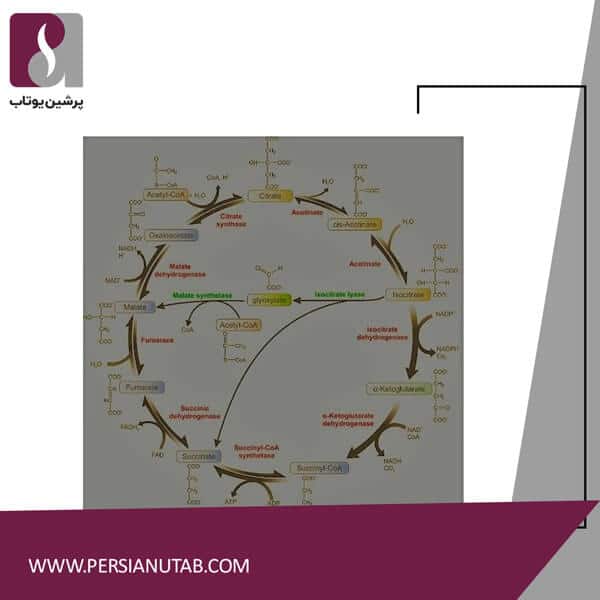
Reaction 3: Hydration to create alcoh
This reaction is a hydration reaction of an alkene to form an alcohol. This water supply does not follow Markovnikov’s rule. The net effect of reactions 2 and 3 is the transfer of the -OH group from C-3 to C-2, which is is cytric acid. This reaction is catalyzed by aconitase.
Reaction 4: Oxidation
This is the first oxidation reaction in which alcohol is converted to ketones. Two hydrogens and 2 electrons are transferred to NAD + to NADH + H +. This is the entry point into the electron transfer chain. The product of this reaction, oxalosuccinic acid, is attached to isocitrate dehydrogenase for the next step. This reaction is catalyzed by isocitrate dehydrogenase.
Reaction 5: Decarboxylation
This is the first step in which a carbon group is destroyed as carbon dioxide in a decarboxylation reaction. The remaining compound now contains 5 carbons and is called alpha-ketoglutaric acid. This reaction is also catalyzed by isocitrate dehydrogenase.
Reaction 6: Oxidation, decarboxylation, thiol ester synthesis
This complex oxidative decarboxylation is driven by three enzymes in much the same way as the formation of acetyl CoA from pyruvic acid. This is actually the only irreversible step in the whole cycle and prevents the cycle from operating in the opposite direction. This is the second oxidation reaction in which alcohol is converted to ketones. Two hydrogens and 2 electrons are transferred to NAD + to NADH + H +. This is another point of entry into the electron transfer chain. This is the second stage in which a carbon group is lost as carbon dioxide in a decarboxylation reaction. Basically, although the carbon is not exactly the same, by the end of this step, the two carbons from the steel CoA have been converted to carbon dioxide. The remaining 4 groups of carbon are attached to CoA by the high-energy bond of thiol ester. Note that the final product, succinyl CoA, contains 4 carbons in the succinate group at the end of the CoA molecule. This reaction is catalyzed by the alpha-ketoglutarate dehydrogenase complex.
Reaction 7: Hydrolysis of Succinyl CoA
ATP synthesis
Thyester bond hydrolysis is associated with the formation of ATP. In fact, guanosine triphosphate is formed first, but is combined with ADP to make more ATP. (This is the only “visible” ATP formed throughout the cycle. Succinic acid, a 4-carbon acid, is the product of this reaction. This is the beginning of the return to the beginning of the cycle. This reaction is catalyzed by succinyl CoA.
Reaction 8: Oxidation
This unusual slightly oxidation reaction results in the removal of hydrogen from saturated alkyl carbon to form an alkene, fumaric acid. The hydrogen receptor is the coenzyme FAD instead of the more common NAD. This will be important when ATP is tabulated from the electron transfer chain, as this coenzyme is complex in the enzyme. Only 2 ATPs are produced from this reaction in the electron transfer chain.This reaction is catalyzed by succinate dehydrogenase.
Reaction 9: Hydration to produce alcohol
It is a simple hydration reaction of an alkene to form alcohol. Place your group where the -OH group is located, as this group must in any case be adjacent to a carboxylic acid group and form malic acid. This reaction is catalyzed by fumarase.
Reaction 10: Oxidation
This is the final reaction in the citric acid cycle. The oxidation reaction of alcohol to ketones is to produce oxaloacetic acid. The coenzyme NAD + transfers two hydrogens and two electrons to NADH + H +. This is the final entry point into the electron transfer chain. This reaction is catalyzed by malate dehydrogenase.
Result
Starting with acetyl Co A with 2 carbon, the citric acid cycle removes these 2 carbons as two molecules of carbon dioxide the Everything about citric acid.




