Sodium citrate formula
What is dosium citrate?
Sodium citrate is a commonly used chemical component in the food industry as a food additive as a flavouring agent or as a preservative. E331 is the most common flavouring agent seen. Sodium citrate is the compound formed after the alkalinizing activity and is the sodium salt of citrate. Upon absorption, sodium citrate dissociates into sodium cations and citrate anions. Here we want to introduce you to the sodium citrate formula.
Properties Of Sodium Citrate
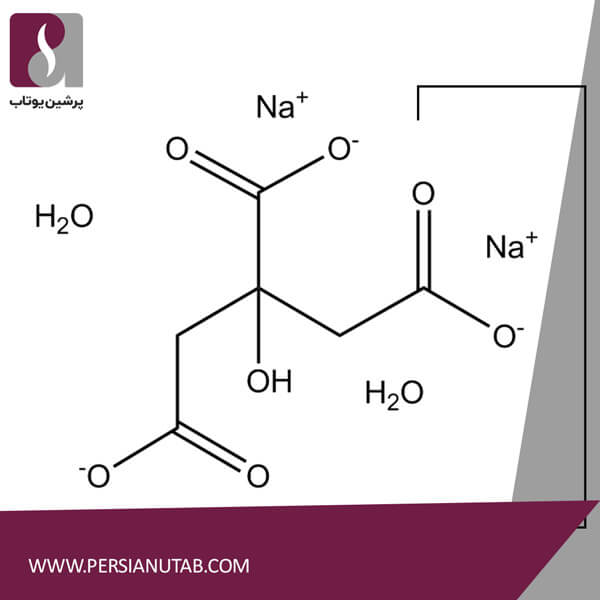
Sodium citrate formula: C6H5O7Na3
Chemical names: Sodium citrate tribasic dihydrate, Citric acid trisodium salt dihydrate and Trisodium citrate.
Sodium Citrate Formula
Sodium Citrate Formula: Sodium citrate is an anticoagulant and hence used in milk-based foods like milk powder, yogurt, jams, sweets, ice cream and many more. Sodium citrate is an organic chemical compound which is found in citric fruits. It is largely used by the food industry as acidifier and flavoring.
We use sodium citrate as a water softener and to adjust the acidity of products. The FDA has deemed the ingredient Generally Recognized as Safe, and Whole Foods has deemed the ingredient acceptable in its body care and cleaning product quality standards. The Cosmetics Ingredient Review has also deemed the ingredient safe for use in cosmetic products. Studies show the ingredient is not a skin irritant or sensitizer.
How Sodium citrate Is Made?
Sodium citrate production occurs by neutralizing citric acid with sodium hydroxide. Citric acid may be produced from fruits or other foods, through yeast fermentation, and by solvent extraction. Most large-scale production occurs by fermenting molasses or other sugar stocks with Aspergillus niger. The liquid is separated by filtration, and the citric acid is separated by precipitation. Sodium citrate is usually offered commercially as the white, crystalline trisodium citrate dihydrate.
Sodium Citrate is the sodium salt of citrate with alkalinizing activity. Upon absorption, sodium citrate dissociates into sodium cations and citrate anions; organic citrate ions are metabolized to bicarbonate ions, resulting in an increase in the plasma bicarbonate concentration, the buffering of excess hydrogen ion, the raising of blood pH, and potentially the reversal of acidosis. In addition, increases in free sodium load due to sodium citrate administration may increase intravascular blood volume, facilitating the excretion of bicarbonate compounds and an anti-urolithic effect.
Applications of sodium citrate
• Sodium Citrate helps in enabling melting of cheeses by not becoming greasy.
• Used in drinks and food as an acidity regulator.
• Used as an emulsifier for oils.
• Used as a pharmaceutical aid.
What does sodium citrate do to blood?
Sodium citrate is the ideal anticoagulant for tests of coagulation, which prevents coagulation by forming a calcium ion complex.
Sodium citrate is the citric acid-based sodium salt. This is often referred to as sour salt, and is primarily used as a food additive.
Can this preparation cause problems?
Recommended doses of sodium citrate are unlikely to cause any side-effects other than making you want to pass a little more urine than normal. If you experience any other symptoms which you think may be due to the medicine, speak with your doctor or pharmacist for further advice this is a Benefits and harms of trisodium citrate.
How to store sodium citrate
• Keep all medicines out of the reach and sight of children.
• Store in a cool, dry place, away from direct heat and light.
Result
Trisodium citrate is a chemical used in the food industry as a flavoring and preservative. The formula of sodium citrate is C6H5O7Na3. This material has many uses. Sodium citrate is also available as a medicine. Be sure to consult your doctor before use.




