Citric acid in the group of carboxylic acids
Citric acid belongs to the group of carboxylic acids.This acid was discovered by the Islamic alchemist Jaber Ibn Hayyan (known as Gabr) in the 8th century, and crystalline citric acid was first isolated from lemon juice in 1784 by the Swedish chemist Karl Wilhelm Schill. Medieval scientists in Europe knew that the crunchy flavor of citrus fruits was due to citric acid. Seventeenth-century naturalists knew that the bite of a red ant was due to the organic acid that the ant injects into the wound. Acetic acid vinegar, formic acid of red ants and citric acid of fruits are all from a family of compounds called carboxylic acids.
Citric acid in the group of carboxylic acids
Citric acid is a carboxylic acid, it can be obtained through various pathways that exist artificially, enzymatically and naturally.These types of organic compounds can be obtained in different ways, some carboxylic acids such as citric acid, lactic acid or fumaric acid are produced by fermentation, most of these carboxylic acids are used in the food industry.Citric acid is harmless, cheap and non-toxic. It has thermal stability up to 175 degrees.This acid is a large carboxylic acid with three ionized hydrogen atoms. It is found in citrus fruits and gives them a sour taste.Ethanoic and citric acids are frequently added to foods to give them a sour taste. Benzoic, propanoic and sorbic acids are used as food preservatives due to their ability to kill microorganisms that lead to spoilage. Methanoic and ethanoic acids are widely used in industry for the production of paints, adhesives and coatings this is a Applications of citric acid.
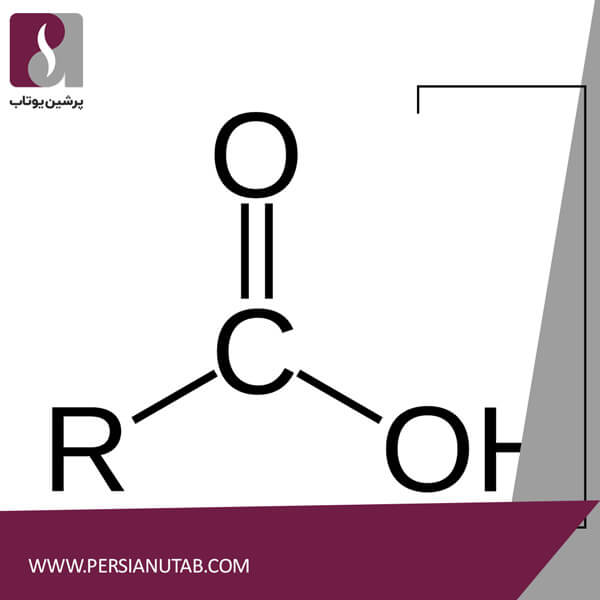
Properties of group carboxylic acids
Carboxylic acids are any group of organic compounds in which a carbon atom (C) is attached by a double bond to an oxygen atom (O) and by a bond to a hydroxyl group (HOH). The fourth bond binds the carbon atom to a hydrogen atom (H) or to another identical compound group.The carboxyl group (COOH) is called the ionyl carbon group (C = O) and the hydroxyl group.The main chemical property of carboxylic acids is their acidity. They are generally more acidic than other organic compounds containing hydroxyl groups, but are generally weaker than inorganic acids.Compounds with excellent chemical and physical properties, special features of this type of organic compounds, their high solubility in polar solvents, such as water, or alcohols, methanol, ethanol and so on.Compounds with excellent chemical and physical properties, special features of this type of organic compounds, their high solubility in polar solvents, such as water, or alcohols, methanol, ethanol and so on.The chemical structure contains carbonyl (-C = O) and a hydroxyl (OH) group, which easily interacts with polar compounds to form H bridges and achieve high boiling points. The carbonyl group (C = O) is one of the functional groups involved in many important reactions.Carboxylic acids are the most important functional group, indicating C = O.Carboxylic acids are versatile organic compounds that can be natural or synthetic, extractable or synthesized, and their chemical structure is highly polar. They are active in organic reactions.
Application of carboxylic acids
Carboxylic acid derivatives, such as alkyl halides, esters and amides, offer different and important applications in different regions.Due to their chemical and physical properties, these types of organic compounds offer countless applications in various fields such as medicine, organic metals, polymers, nanotechnology, food, etc.Carboxylic acids are primarily used in the manufacture of soaps, detergents, medicines, plastics, dyes, fabrics, perfumes and animal feed.The production of beverages and many other food products involves the use of many carboxylic acids.Rubber production involves the use of acetic acid as a coagulant.Carboxylic acids have numerous applications in the rubber, textile and leather industries.Ethylene diamine tetraacetic acid is a widely used chemical.The synthesis of many drugs involves the use of these compounds. Therefore, carboxylic acids are very important in pharmacy.The production of many polymers involves the use of compounds containing the carboxyl functional group.
Carboxylic acids market
The growth of the global carboxylic acid market is expected to increase with the increasing demand for vinyl acetate monomer or VAM, due to the increasing demand for vinyl ester resins used to cover the surfaces of materials used for storage and packaging of food and beverages. Vinyl esters, in turn, are produced by the reaction of acetic acid with a vinyl acetate monomer (resin). The increase in demand for packaged food will have a significant impact on the market in the foreseeable future due to the increase in global GDP as well as the lack of time in double-income households. In addition, demand for ethylene-vinyl acetate (EVA) is expected to increase due to its applications in the production of adhesives used in food packaging. The global VAM market size was over $ 7.4 billion in 2015 and is expected to grow by nearly 4.5% CAGR from 2016 to 2024. Rising demand for cosmetics and personal products also catalyzes global market demand for carboxylic acid. Excessive use of citric, stearic, butyric and valeric acids in the manufacture of these products will affect the global carboxylic acid market in the forecast period. Increasing public awareness of cosmetics, in addition to increasing usable revenue, will significantly increase the size of the market in the coming years. The use of acids for sterilization to prepare perfumes and prevent germs has a dominant effect on the market. More than 80% of these products are personal care products for cosmetics, skin care products, perfumes and hair care products. Another important enhancer in the carboxylic acid market is the increasing use of organic acid in the animal feed industry to prevent the growth of microbes. Carboxylic acids such as formic acid and propionic acid inhibit the growth of Salmonella, which contaminates food and causes animals as well as humans. More than 45% of the global livestock feed market is for chickens and more than a quarter is used for pigs. Increasing the consumption rate of poultry and pigs for their beneficial protein content is likely to increase the market size of carboxylic acid as a feed additive. Regulations banning antibiotic growth enhancers in animal feed, especially for poultry, pigs and cattle, will also have a positive effect on the industry. However, the availability of organic and natural alternatives in the feed industry will hinder the size of the carboxylic acid market in the coming years, as they contribute to probiotics, amino acids, nutrients, antimicrobials and antioxidants. And several other newly developed alternatives have been lost. Regulations on the production of petroleum-based carboxylic acids with long-chain hydrocarbons also curb the growth of the global carboxylic acid market in the forecast period.




